Traumatic brain injury is the leading cause of death and disability in children and young adults in the United States. It is also a major concern among people over 75, with high rates of death and hospitalization.1 Common mechanisms of injury over all age groups include motor vehicle crashes, falls, injuries from firearms, and assaults. It has been a prevailing thought that there is little that prehospital care providers can do for patients with traumatic brain injuries, but recent studies and improved protocols reveal that EMS has a considerable impact on positive patient outcomes.
Case Introduction
EMS responds to a 28-year-old male who fell off his bicycle, and was not wearing a helmet. A passer-by found the cyclist semi-conscious, laying on the road for an unknown period. He assisted the cyclist to his feet and then sat him on the front seat of his car. Initial assessment reveals: Airway – patent with large amounts of vomit observed on the patient’s chin, mouth, shirt and on the ground by his feet; C Spine – potentially compromised, due to a suspected traumatic mechanism of injury; Breathing – regular rhythm and depth, slightly labored; Pulses present in all four extremities. The patient is now experiencing periods of lucidity, alternating with response only to voice.
When awake, the patient states he has no allergies, took his insulin and Lipitor today, and has a history of diabetes and high cholesterol. He states he was riding for an hour, gradually becoming dizzy and weak, and then thinks he passed out and crashed. The patient says he woke up laying on the road with someone hovering above him, asking if he was okay. There is no damage noted to the bike suggesting a collision with a vehicle. Vitals: pulse 98, ventilations 24, SpO2 98%, BP 138/90, BGL 45 mg/dL, GCS 13 (E3, V4, M6).
Physical exam reveals a large hematoma on the right parietal/temporal area and crepitus when the cranium is palpated; pupils are equal but react sluggishly; abrasions on the right side of his face, no drainage observed from the nose or ears; posterior neck painful when palpated (no deformity), no jugular venous distention observed; abrasions on lateral right chest and arm; lung sounds are clear throughout, no palpated chest deformity; abdomen is soft and non-distended; pelvis is intact with slight pain upon palpation of right iliac crest; abrasions observed on the right lateral thigh and leg; distal pulses present – movement and sensation intact in all four extremities. Exam of the posterior trunk and legs is unremarkable except for abrasions. A cervical collar is applied and a scoop stretcher is carefully placed under the patient. He is then moved onto the ambulance cot for transport.
Primary and Secondary Traumatic Brain Injury (TBI)
There are two distinct categories – primary and secondary TBI. Primary traumatic brain injury occurs during an initial impact when physical structures of the brain are displaced, often resulting in lacerated vessels, mechanical disruption of brain cells and increases in vascular permeability.3 When the skull and brain are pierced, this is called a focal injury, and when an external force produces excessive movement of the brain within the skull, this is called a diffuse injury. Primary injury may be a skull fracture, epidural, subdural or intracerebral hematoma, coup and contrecoup contusions, concussion, impaled object, or penetrating injury from a bullet, knife or shrapnel.
Secondary traumatic brain injury furthers the cellular brain damage from the effects of a primary injury. This typically develops over a period of hours or days following the initial head trauma. Common mechanisms that lead to secondary brain injury are breakdown of the blood-brain barrier – causing cerebral edema, hypoxia, hypotension, hypercarbia, and increased intracranial pressure. Many of these secondary injuries may be preventable with early and effective airway management, ensuring good ventilation and oxygenation, and maintaining blood pressure.
The Case Continues
The patient has a confirmed history of diabetes and has an altered LOC, so the EMS crew decides to administer dextrose to increase his serum glucose level. Cautious, due to the potential head injury, 12.5 grams is given IV. This increases the serum level to 106 mg/dL, but the patient suddenly begins to experience a tonic-clonic seizure. He begins forcefully vomiting and loses consciousness. The EMS crew carefully log rolls the patient onto his left side, while maintaining in-line positioning, and suctions his airway. Five milligrams of IV diazepam are administered, which stops the seizure. He still maintains a partially intact gag reflex, but his SpO2 has decreased to 93%. A nasopharyngeal airway is inserted into the left nare, and oxygen is administered via non-rebreather mask flowing 15 Lpm. Repeat vitals: Ventilations 28; Pulse 106 regular with diminished pedal pulse strength, BP 146/88, BGL 104 mg/dL; SpO2 98% with oxygen; GCS 10 (E2, V3, M5). The hematoma has not appeared to have grown in size, but the patient’s right pupil is larger when compared to the left and has become less reactive.
How does a TBI affect the brain?
Blood, cerebrospinal fluid and brain tissue all exist in a fixed ratio within the head. The brain takes up 80% of the cranium, 10% is blood within the vessels and 10% is cerebrospinal fluid. Altogether, these exert a certain amount of force, known as intracranial pressure (ICP). If there is an increase in volume of any one of these three components, one or both of the others must decrease volume in order to maintain ICP. Various events that can increase ICP include brain tumors, bleeding within the brain, meninges and/or cranium, and cerebral edema.
If one or more of the three cranial components decrease in volume from increased ICP, this also compromises cerebral perfusion pressure (CPP). Cerebral perfusion pressure is the pressure required to ensure blood flow to the brain. It is dependent upon intracranial pressure remaining constant as well as a sufficient mean arterial pressure (MAP). MAP is a measure of an average pressure exerted in the arteries during a single cardiac cycle – regulated by adjustments in cardiac output and systemic vascular resistance. Normal range for MAP is 70-110 mmHg, and normal range for ICP is 0-15 mmHg. Any change in either MAP or ICP will affect brain perfusion.
An adequate CPP is between 70-90 mmHg. When cerebral perfusion pressure drops below 70 mmHg for even short periods, cerebral hypoxia and ischemia can occur. The brain prevents a dangerous CPP drop via autoregulation. It is an automatic response that reacts to fluctuations in the systemic arterial pressure with an opposite reaction by the cerebral vessels. For example, an increase in systemic blood pressure causes the brain to constrict the cerebral arteries to limit cerebral blood volume and any increase in intracranial pressure. Conversely, if the patient’s systemic blood pressure decreases, the cerebral vessels dilate, allowing for increased blood flow and maintenance of brain perfusion.3
Autoregulation is effective when the MAP is between 60-130 mmHg and when ICP is less than 40 mmHg. If the MAP rises above 130 mmHg, the cerebral vessels leak fluid into surrounding brain tissue and increase ICP. If the MAP drops below 60 mmHg, cerebral blood flow is insufficient and causes ischemic tissue injury. When ICP exceeds 40 mmHg, autoregulation is disrupted. The maintenance of cerebral blood flow now relies upon the brain being able to increase systemic blood pressure by peripheral vessel vasoconstriction. This does increase/maintain blood flow to the head, but forces a greater-than-normal volume of blood into the cranium. The cerebral vessels remain dilated due to autoregulation loss, resulting in a progressive increase in cerebral edema, intracranial pressure, and brain ischemia.
The Case Continues
The patient is loaded into the ambulance for a 10-minute transport to the trauma center. Seizure activity does not return, and he appears postictal. In an attempt to assess mental status, the EMT applies a vigorous sternal rub, which causes the patient to moan loudly and flail his right arm away from midline. The EMT also observes the patient’s breathing progressively gets faster, then slows, stops, then repeats the cycle. The NRB mask is removed and the EMT begins to ventilate the patient with a BVM and oxygen. The patient does not resist the assisted ventilatory support and accepts insertion of an oropharyngeal airway. Repeat vitals: Pulse 80 regular, absent pedal pulses and diminished radial pulse strength; BP 162/70; Ventilations via BVM at 18/minute, SpO2 99%; BGL 100 mg/dL; GCS 7 (E1, V2, M4). A repeat physical exam reveals no changes except the right pupil is further dilated and is not reactive to light. The patient is positioned supine on the ambulance cot and rapid transport is initiated.
Signs and Symptoms of Increased ICP and Herniation
The best indicator of increasing ICP is a changing mental status.4 These changes can range from restlessness and confusion to irritability, combativeness and coma. Other indicators include severe headache, projectile vomiting, personality changes, slowing of speech, loss of reflexes, abnormal posturing, seizure activity, pupillary changes, abnormal breathing patterns, bradycardia and progressively increasing hypertension with an associated widening pulse pressure. These last three findings constitute the Cushing reflex, which is a primary sign of brainstem herniation.
As ICP increases, eventually the cranial vault completely fills. If ICP continues to increase further – continuing the expansion of contents, they are forced toward the only place in the skull that has a lower pressure, the foramen magnum. Anatomically, the brainstem is sandwiched between the expanding cerebrum, blood and CSF superiorly and the inferior foramen magnum. As ICP increase continues, the brainstem progressively gets compressed, and herniation begins.
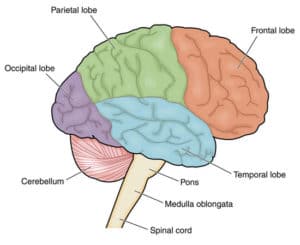
The midbrain is affected first. Autoregulation is lost by this point, so the brain attempts to increase systolic blood pressure to sustain a MAP that can maintain CPP. The increased blood flow to the brain compounds the ICP buildup, eventually compressing the expanding contents onto the midbrain and overstimulating the vagus nerve. The patient’s heart rate decreases from the massive acetylcholine interaction, and bradycardia develops. This leads to further complications of maintaining CPP, due to an associated cardiac output drop. Cheyne-Stokes breathing pattern manifests along with an abnormal pain flexor response (decorticate posturing).
As ICP continues to increase, the midbrain is disabled and the pons now must try to compensate. Think of the pons as 2 pieces – a superior half and inferior half. The superior half is the home of the acceleration center. Overstimulation from ICP leads to the respiratory pattern acutely changing from Cheyne Stokes into central neurogenic hyperventilation. Once the superior pons is crushed and disabled, the inferior portion is overstimulated – home to the deceleration center. The patients’ breathing acutely becomes irregular in depth, rate and rhythm, Bradycardia worsens, pulse pressure continues to widen, and the patient’s blood pressure continues to rise. Systolic blood pressure can easily climb into the 200-250+ mmHg range. Unilateral or bilateral sluggish/inactive pupils may be observed, and painful stimulus produces an abnormal extensor response (decerebrate posturing).
When the midbrain and the pons’ functions are destroyed, the remaining active portion of the brainstem is the medulla oblongata. Its vegetative functions of maintaining respiration and circulation are rapidly compromised by the crushing force of the brain mass. Systemic blood pressure regulation is lost and the patient becomes profoundly hypotensive, resulting in the loss of cerebral perfusion. The patient’s breathing pattern becomes agonal, then apneic. Bradycardia decompensates into cardiac arrest in many cases. The pupils become fixed and dilated and there is neither an extensor nor a flexor response to painful stimulation. Such findings typically suggest a level of ICP that is not survivable.
Management Pearls
The prehospital management of increased ICP centers on maintaining and protecting the airway, breathing, and circulation in an attempt to control ICP and prevent a decrease in cerebral perfusion. A study published in JAMA Surgery: The Journal of the American Medical Association5highlights the importance of treating and preventing the three “H Bombs” (Hypoxia, Hypotension and Hyperventilation) that lead to irreversible brain damage and death following a brain injury. This study stems from a project known as EPIC: Excellence in Prehospital Injury Care.
Hypoxia
Be sure to oxygenate any TBI patient to maintain an SpO2 greater than 90% at all times, as even one episode of hypoxia can double the mortality of a head-injured patient.6 Ideally, administer oxygen by the most appropriate method, to obtain an SpO2 close to 100%. Pay particular attention when placing an advanced airway and the time it may take to do so. Prolonged and/or multiple airway insertion attempts may lead to rapid oxygen desaturations and increase the chance for hypoxia-related complications.
Hypotension
A single episode of hypotension is very detrimental to ischemic brain tissue and cerebral blood flow. Any episode of hypotension also doubles the mortality from head injuries.6 If the patient is, or becomes hypotensive, infuse isotonic IV fluid to establish and maintain a systolic blood pressure of at least 90 mmHg. However, do not delay transport to establish an intravenous line in the field.
Hyperventilation
First responders have always been taught to hyperventilate patients with suspected TBI. However, recent research has shown that while hyperventilation lowers intracranial pressure in some instances, more often it deprives the brain of blood and oxygen due to the extreme reduction of serum carbon dioxide. The cerebral vasoconstriction that results causes the brain insult. Hyperventilation is not necessary with head-injury patients who do not have signs or symptoms of herniation, as their auto regulatory mechanism is still intact. EPIC recommends that TBI patients never be hyperventilated, even if herniation is suspected. It is a very rare event in the prehospital setting – what is observed as possible herniation, most times is not the case. When ventilating a patient suspected of having a TBI, the rate be should 10/minute for patients older than 15, 20/minute for patients 2 – 14 years old and 25/min for patients less than 2 years old. Each ventilation should be over one second and enough to initiate chest rise. Capnometry should be monitored throughout care and maintained between 35-45 mmHg. The EPIC results recommend the magic number be no higher than 40 mmHg. A normal capnograph waveform should be present as well. If not, assess for issues that may have altered the shape, angle or plateau of the waveform, as these sometimes contribute to patient decline.
Additionally, consider these points for a patient with suspected TBI. One, the severity of a traumatic brain injury, according to Glasgow Coma Score, is graded as: Mild 13-15, Moderate 9-12, and Severe 3-8.2 The lower the score, the worse the prognosis and lessened ability for a patient to maintain their airway. Two, employ spinal motion restriction protocol, as warranted. Three, always consider rapid transport by the most-appropriate means to a trauma center capable of neurosurgery. Four, if a seizure occurs, administer a benzodiazepine to stop the seizure activity. Be careful with the dosage, however, so as not to induce hypotension. Post-seizure, check for reduced blood glucose levels and manage accordingly. Lastly, maintain a patient’s normal body temperature by the most appropriate method.
Case Conclusion
The paramedic decides to intubate the airway, due to the patient’s LOC being severely diminished and no gag reflex present. The tube is passed without difficulty or delay, secured, and the BVM is reattached along with in-line capnometry. Observing signs and symptoms of a traumatic brain injury and possible brainstem herniation, the EMT slows ventilations to 10/minute. He gives enough volume for observable chest rise, and maintains an EtCO2 between 35-40 mmHg. Remaining transport time to the hospital is five minutes. Repeat vitals: Pulse 75 regular with a thready radial pulse; BP 178/62; SpO2 99%; GCS 5 (E1, V1, M3). Another peripheral IV line is established, run at KVO rate and the cardiac monitor is applied – showing normal sinus rhythm. The hematoma appears to have increased in size and the right pupil is fixed and dilated. The remainder of the transport is unremarkable and full report is given to the receiving ED staff without incident. The patient is rushed to surgery where a large epidural bleed on the temporal area of the patient’s brain is evacuated and repaired.
References
- Traumatic Brain Injury in the United States Fact Sheet. Centers for Disease Control and Prevention, National Center for Injury Prevention and Control, Division of Unintentional Injury Prevention. January 12, 2015.
- Dawodu, S.T. (2019, June 27). Traumatic Brain Injury (TBI) – Definition, Epidemiology, Pathophysiology. Retrieved from: https://emedicine.medscape.com/article/326510-overview#a1
- Duncan, T., et.al. (2007, July 1) Beyond the Basics: Brain Injuries. Retrieved from: https://www.emsworld.com/article/10321801/beyond-basics-brain-injuries
- Snyder, S. (2012, February 6). Troubled Mind: The Lowdown on Increased ICP. Retrieved from: https://www.emsworld.com/article/10624283/troubled-mind-lowdown-increased-icp
- Bassett, J (2019, July 1). Retrieved from: https://www.emsworld.com/article/1222954/avoiding-three-h-bombs-prehospital-tbi-management
- Duckworth, R (2018, March 26) EPIC: An EMS-centered approach to head injuries. Retrieved from: https://www.ems1.com/ems-training/articles/378624048-EPIC-An-EMS-centered-approach-to-head-injuries/